Decompression models, by design, track the inert gas dissolved throughout the body and ensure that it doesn't exceed a specific threshold upon ascent (this threshold is sometimes called M-value, or MPTT - Maximum Permissible Tissue Tension, or something else depending on the decompression model used).
To calculate the diffusion of the inert gas throughout the body, decompression scientists use a simplified model of human respiratory physiology. In essence, this model considers four different zones to take into consideration when calculating the transition of the inert gas from one environment to the other:
- The gas in the blend: present in the cylinder/atmosphere, this is the gas that will be in contact with the mouth of the diver
- The inspired gas: this is the gas found in the upper airways. As the dry gas gets humidified from the saturated water vapor present, the partial pressure of the inert gases drops slightly.
- The alveolar gas: this is the gas found in the alveoli. In this environment, on top of the saturated water vapor pressure, we will have to take into account the production of carbon dioxide and the metabolism of oxygen.
- The gas dissolved in the tissues/compartments: this is the gas that is used by decompression models to limit supersaturation and to calculate decompression stops.
To calculate the partial pressure of the inert gases in the tissues, we will first have to calculate the gas content of the first three "zones": the transfer of gases between each of those zones is assumed to be instant, while the transfer of gas between the alveolar and compartmental gas follows a first-order kinetic rule. Calculating the dissolved inert gas partial pressures in the tissues will require us to solve a differential equation whose solution will look something like this:
\[ p_i(t) = p_0e^{-k_i(t-t_0)} + k_i \int_{t_0}^{t} p_{alv}(s)e^{k_i (s-t)} ds \]The alveolar pressure will need to be defined to finish solving this equation. In any case, the scope of this article is not to cover the interaction between alveolar gas and compartmental gas (as it is a vast topic of its own, which could have multiple articles written on it), but rather how to obtain an expression of inert alveolar gas.
The Alveolar Environment
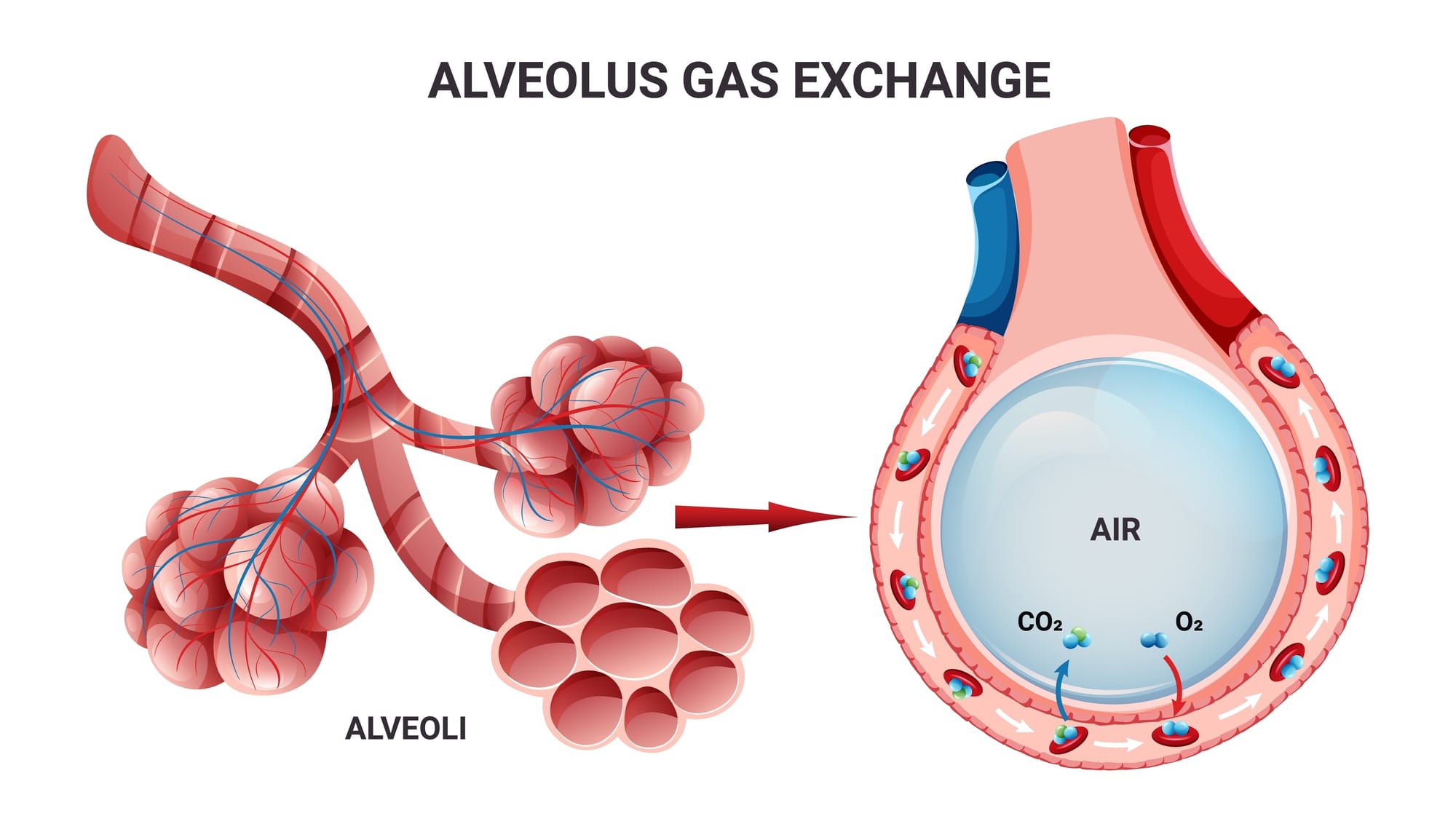
The alveoli are small gas-holding sacs in the lungs and are the sites of gas exchange between the blood and the air. To model the gas environment properly, decompression scientists make a few assumptions about the alveoli:
- The pressure in the alveoli is considered to be equal to the ambient pressure.
- The total pressure inside the alveoli is equal to the sum of the gases inside it:
With the alveolar carbon dioxide pressure and saturated water vapor considered to be constant and independent of depth. Namely, we will consider:
\[p_{a,CO_2} = 40 \text{ mmHg} \approx 0.05333 \text{ bar} \\ p_{H_2O} = 47 \text{ mmHg} \approx 0.06266 \text{ bar}\]- As oxygen is metabolized inside the body, the alveolar oxygen is lower than the inspired oxygen and follows the alveolar equation:
With R being the respiratory quotient, which is defined as the volume of carbon dioxide produced over the volume of oxygen consumed. Its value varies depending on the decompression model used, and ranges from 0.7 to 1.
- The inspired oxygen is humidified and loses some pressure to the saturated water vapor:
- The alveolar inert gases are considered to be in the same proportion to each other as the inert gases found in the inhaled blend:
Alveolar inert gases
We will now express the alveolar partial pressure of an inert gas as a function of its fraction in the mix and the ambient pressure. For decompression models, there are two scenarios to take into account: constant ambient pressure (if the diver is stationary) or variable ambient pressure (if the diver ascends or descends). In the latter case, we consider the diver to move at a constant rate. We will consider both cases below.
If the diver is using a Closed Circuit Rebreather, there are more cases, but this article will focus on constant gas fractions and an open-circuit setup.
Constant ambient pressure
For a constant ambient pressure, the alveolar inert gas partial pressure will also be constant, and solving the differential equation will give us the so-called Haldane equation.
We can start by using the formula expressed in the second assumption:
\[ p_{alv}=p_{a,O_2}+p_{a,CO_2}+p_{H_2O}+\sum{p_{a, inert}}\\ \Leftrightarrow \sum{p_{a, inert}} = p_{alv} - (p_{a,O_2}+p_{a,CO_2}+p_{H_2O})\\\\ \]\[ \text{Assuming } p_{alv} = p_{amb} \]
\[ \sum{p_{a, inert}} = p_{amb} - (p_{a,O_2}+p_{a,CO_2}+p_{H_2O})\\ \]
\[ \text{Substituting } p_{a,O_2} \]
\[ \sum{p_{a, inert}} = p_{amb} - (p_{i,O_2} - \frac{p_{a,CO_2}}{R} + p_{a,CO_2} \cdot f_{i,O_2} \cdot \frac{1-R}{R}+p_{a,CO_2}+p_{H_2O})\\ \]
\[ \text{We define } F = p_{a,CO_2} \cdot f_{i,O_2} \cdot \frac{1-R}{R}\\ \]
\[ \begin{aligned} \sum{p_{a, inert}} &= p_{amb} - (p_{i,O_2} - \frac{p_{a,CO_2}}{R} + F+p_{a,CO_2}+p_{H_2O})\\ &= p_{amb} - ((p_{amb} - p_{H_2O})f_{O_2} - \frac{p_{a,CO_2}}{R} + F+p_{a,CO_2}+p_{H_2O})\\ &= p_{amb} - (p_{amb} - p_{H_2O})f_{O_2} - (-\frac{p_{a,CO_2}}{R} + F+p_{a,CO_2}+p_{H_2O})\\ &= p_{amb} - (p_{amb} - p_{H_2O})(1-\sum{f_{inert}}) - (\frac{R-1}{R}p_{a,CO_2}+p_{H_2O}+F)\\ &= p_{amb} - p_{H_2O} - (p_{amb} - p_{H_2O})(1-\sum{f_{inert}}) - (\frac{R-1}{R}p_{a,CO_2}+F)\\ &= (p_{amb} - p_{H_2O})(1 - 1 + \sum{f_{inert}}) - (\frac{R-1}{R}p_{a,CO_2}+F)\\ &= (p_{amb} - p_{H_2O})\sum{f_{inert}} - (\frac{R-1}{R}p_{a,CO_2}+F)\\ \end{aligned} \]
\[ \text{We can substitute } F \text{ back in the right parenthesis:}\\ \]
\[ \begin{aligned} \frac{R-1}{R}p_{a,CO_2}+F &= \frac{R-1}{R}p_{a,CO_2}+ p_{a,CO_2} \cdot f_{i,O_2} \cdot \frac{1-R}{R}\\ &= \frac{R-1}{R}p_{a,CO_2} - p_{a,CO_2} \cdot f_{i,O_2} \cdot \frac{R-1}{R}\\ &= \frac{R-1}{R}p_{a,CO_2}(1-f_{i,O_2})\\ &= \frac{R-1}{R}p_{a,CO_2}\sum{f_{inert}} \end{aligned} \]
\[ \text{Which allows us to simplify:}\\ \]
\[ \begin{aligned} \sum{p_{a, inert}} &= (p_{amb} - p_{H_2O})\sum{f_{inert}} - (\frac{R-1}{R}p_{a,CO_2}+F)\\ &= (p_{amb} - p_{H_2O})\sum{f_{inert}} - \frac{R-1}{R}p_{a,CO_2}\sum{f_{inert}}\\ &= (p_{amb} - p_{H_2O} - \frac{R-1}{R}p_{a,CO_2})\sum{f_{inert}} \end{aligned} \]
\[ \text{Substituting the formula from the 5th assumption on the left hand side:}\\ \]
\[ \begin{aligned} \frac{p_{a, i}\sum{f_{inert}}}{f_i} &= (p_{amb} - p_{H_2O} - \frac{R-1}{R}p_{a,CO_2})\sum{f_{inert}}\\ \Leftrightarrow p_{a, i} &= (p_{amb} - p_{H_2O} - \frac{R-1}{R}p_{a,CO_2})f_i \end{aligned} \]
Finally, we can define the full inert gas alveolar equation to be:
\[ \boxed{p_{a, i} = (p_{amb} - p_{H_2O} - \frac{R-1}{R}p_{a,CO_2})f_i} \]Some models (such as the Bühlmann ZH-L models) will set R=1, which will result in the right term being completely ignored, and to only take the water vapor pressure into account.
Variable ambient pressure
In this scenario, we introduce a variable r, describing the speed of the diver. By convention, r is positive upon descent and negative upon ascents. Instead of the ambient pressure being constant, it can now be defined as:
\[ p_{amb}(t) = \rho gr(t-t_0)+ p_{amb}({t_0})\\ \]\[ \begin{aligned} \rho &\text{ Describing water density}\\ g &\text{ Describing gravitational acceleration}\\ t-t_0 &\text{ Describing the elapsed time}\\ p_{amb}({t_0}) &\text{ Describing the ambient pressure at the start of the movement} \end{aligned} \]
For simplicity, we will consider each movement as segments and set t0 = 0 with t describing the total elapsed time of that movement. The equation then simplifies to:
\[ p_{amb}(t) = \rho grt+ p_{amb}(0)\\ \]We can start from the inert gas alveolar equation:
\[ \begin{aligned} p_{a, i} &= (p_{amb} - p_{H_2O} - \frac{R-1}{R}p_{a,CO_2})f_i\\ \Leftrightarrow p_{a, i}(t) &= (p_{amb}(t) - p_{H_2O} - \frac{R-1}{R}p_{a,CO_2})f_i\\ &= (\rho grt+ p_{amb}(0) - p_{H_2O} - \frac{R-1}{R}p_{a,CO_2})f_i\\ &= \rho grtf_i + (p_{amb}(0) - p_{H_2O} - \frac{R-1}{R}p_{a,CO_2})f_i\\ \end{aligned} \]\[ \text{We will define }Q = \rho grf_i \text{ as the rate of change of the inspired inert gas. We then have:} \]
\[ \boxed{p_{a, i}(t) = Qt + p_{a, i}(0)} \]
Examples
- What is the alveolar pressure of nitrogen in a human breathing air at the surface at sea level ? By considering pamb=1.01325 bar, fN2 = 0.79, and R=0.8) We can calculate:
- For the second example, let's illustrate the alveolar pressures in a Bühlman-based model, which considers R=1. What is the alveolar pressure of nitrogen and helium in a human breathing a trimix 20/35 blend at a depth of 50 meters? In this scenario, we will consider the ambient pressure to be pamb=6.0409 bar, fN2 = 0.45, fHe = 0.35. We can calculate: